A different brand of iron tablet was analysed by Hitration with 0.0093 mol.L potassium dichromate via the
Por um escritor misterioso
Descrição
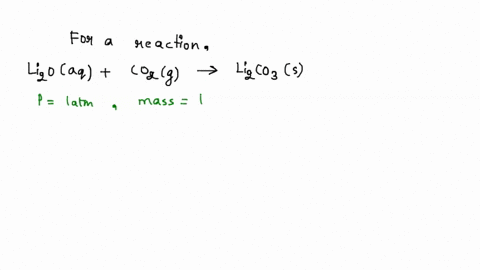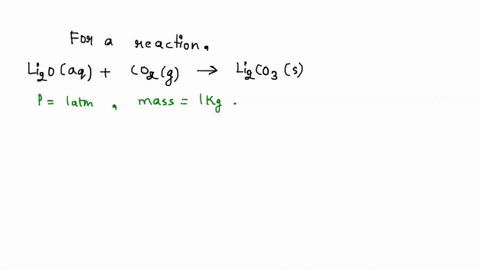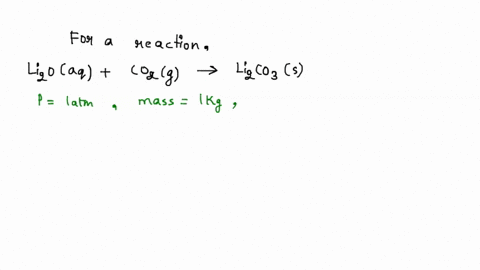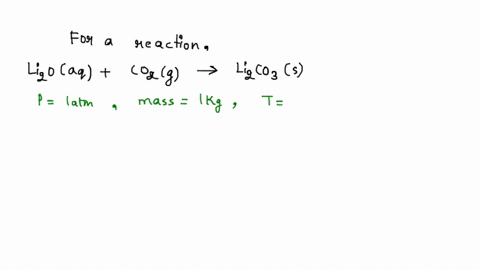
SOLVED: Iron supplement tablets contain iron (II) ions. The amount of iron present can be found by carrying out a redox titration with potassium permanganate in acid conditions. The redox reaction is

What amount of CuSO4.5H2O is required for the liberation of 2.54 g of I2 when it titrates with KI? - Quora

Food Science and Tecnology, volume 35, number 1, 2015 by Editora Cubo - Issuu

Energy-Efficient and Green Extraction of Bismuth Metal in Methanesulfonic Acid-Based Membrane Electrochemical Systems

PDF) Test strip for determination of nitrite in water

Enhanced phytoremediation of Metal(loid)s via spiked ZVI nanoparticles: An urban clean-up strategy with ornamental plants - ScienceDirect

PDF) Modern Analytical Chemistry

PDF) Analytical-Chemistry Laura G Anzaldo

Chemistry neet

A2 Expt 14.4 (8) Analysis of Iron Tablets, PDF, Iron

Powdered Potassium Dichromate Standard MERCK 80 Gm., Grade: Analytical

What amount of CuSO4.5H2O is required for the liberation of 2.54 g of I2 when it titrates with KI? - Quora
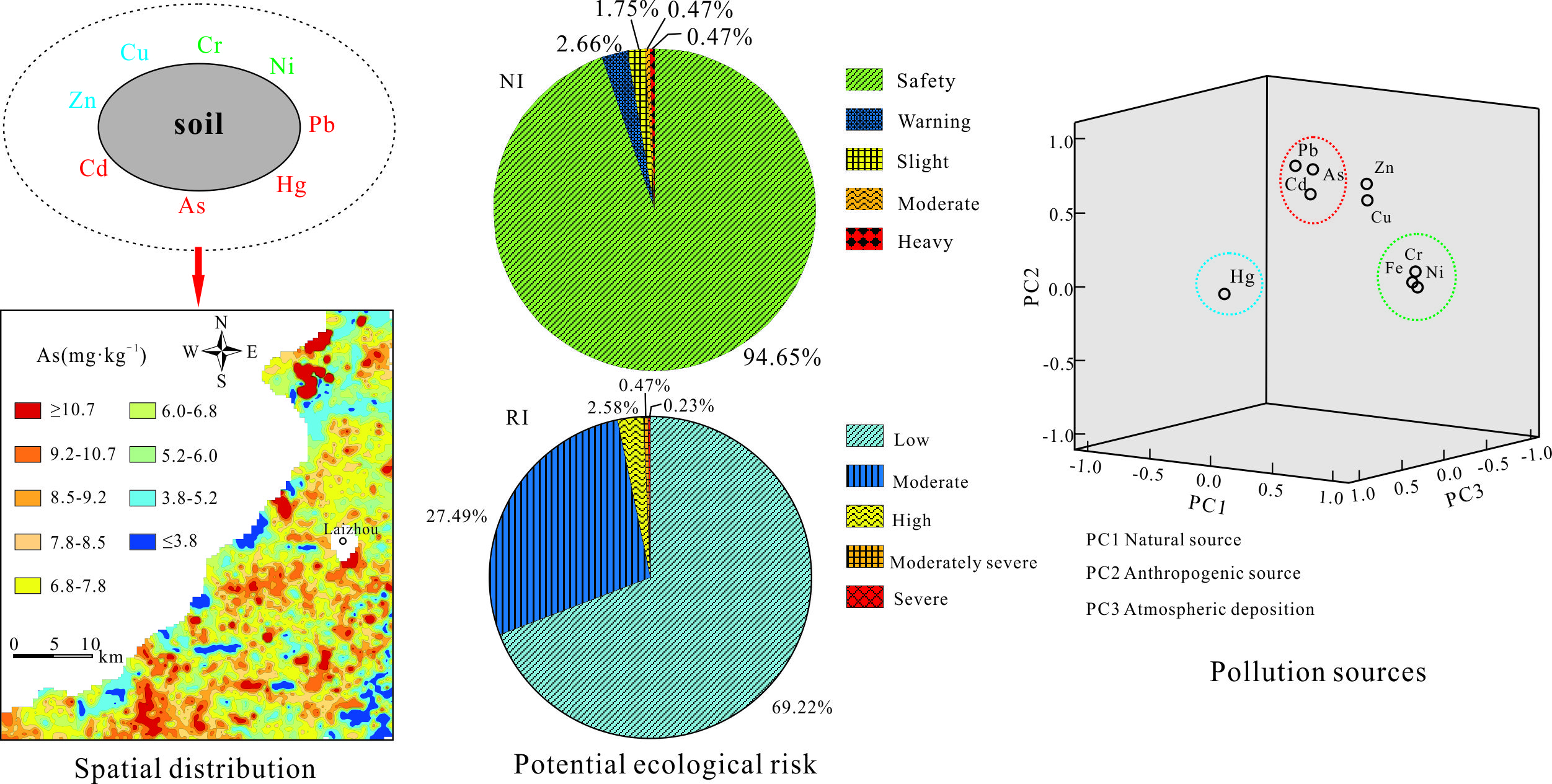
Water, Free Full-Text

23.4 Variable Oxidation States of Transition Elements Flashcards

Solved Potassium dichromate in acidic solution is used to
de
por adulto (o preço varia de acordo com o tamanho do grupo)