FDA allows Houston cancer doctor to resume drug trial
Por um escritor misterioso
Descrição
Federal regulators have lifted a partial hold on a clinical trial performed by Stanislaw

Texas Medical Board sanctions controversial cancer doctor Burzynski

Pioneering' breast cancer research yields more dollars in race for treatment, solutions - University of Mississippi Medical Center

When Cancer Patients Ask About Weed, Many Doctors Say Go For It – Houston Public Media
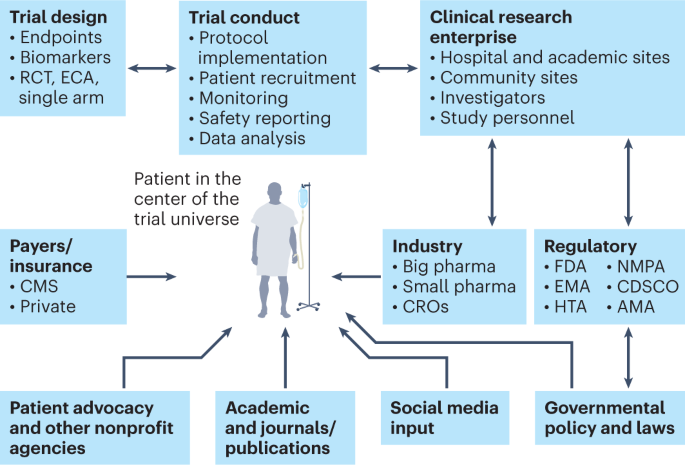
The next generation of evidence-based medicine

Penn Medicine's Dr. Carl June to chair Nanjing Bioheng Biotech's scientific advisory board - Philadelphia Business Journal
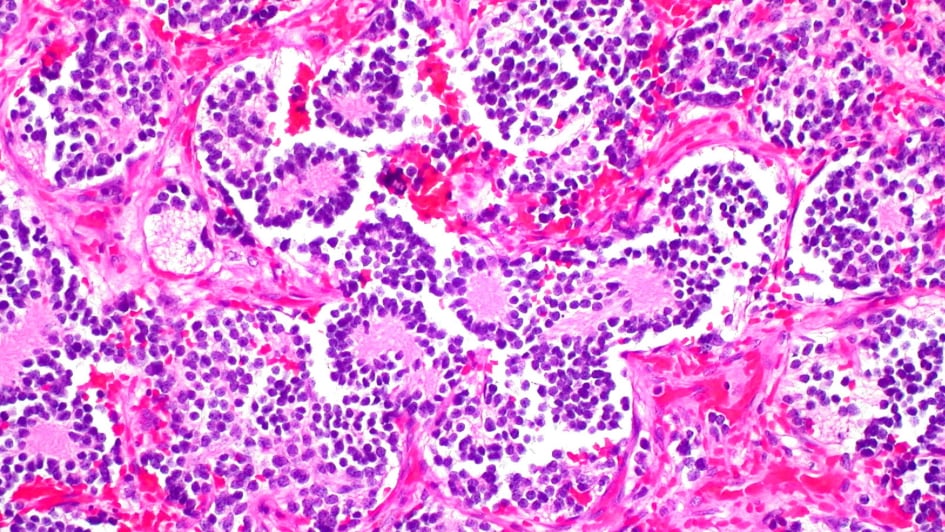
New drug targeting high-risk children's cancer is ready for trials - The Institute of Cancer Research, London

Physician Resume Samples

Intravesical nadofaragene firadenovec gene therapy for BCG-unresponsive non-muscle-invasive bladder cancer: a single-arm, open-label, repeat-dose clinical trial - The Lancet Oncology
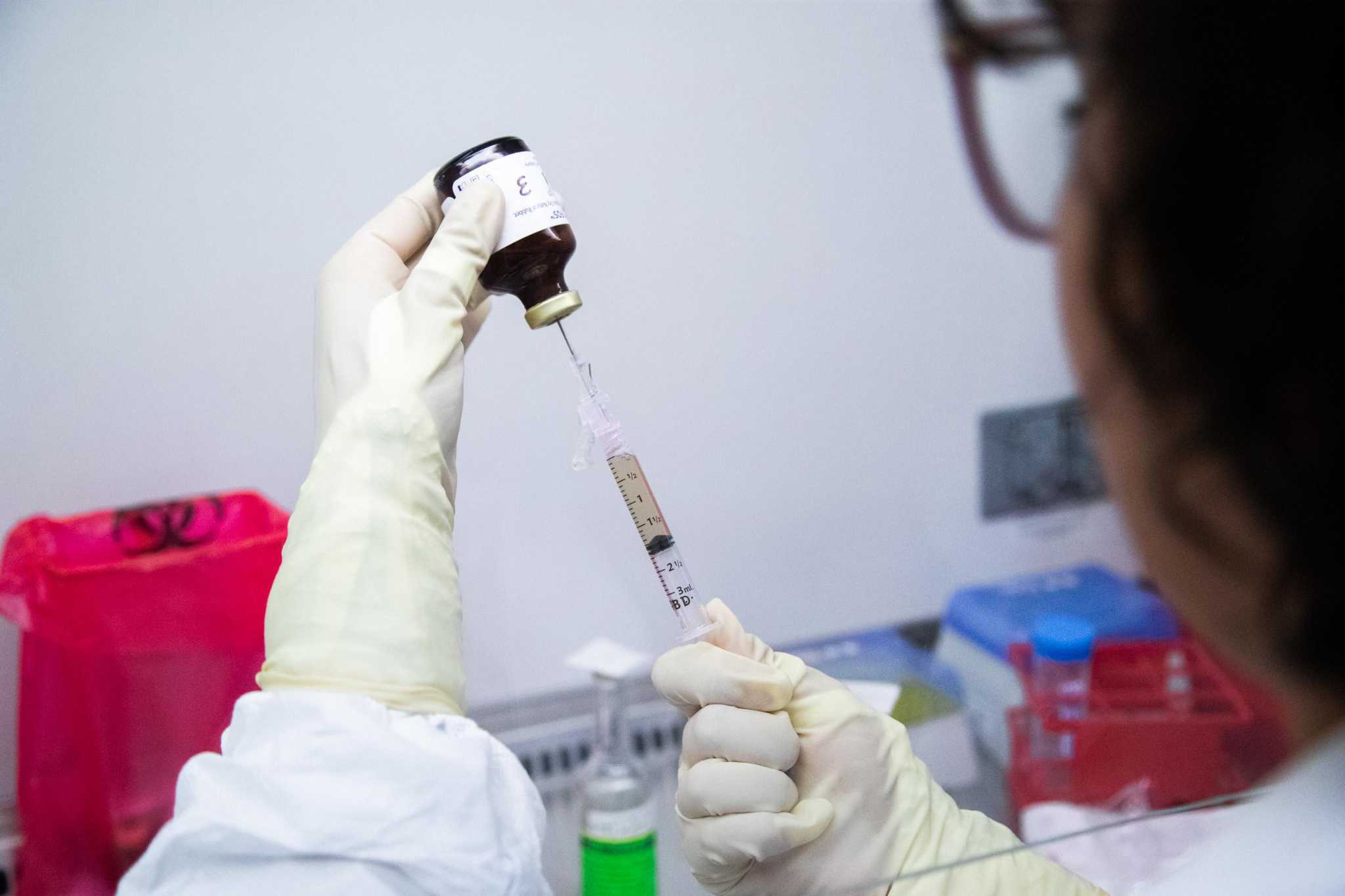
FDA clears MD Anderson to test skin cancer treatment on humans

Doctor claims to cure pediatric cancer, critics skeptical

Shubham Pant MD Anderson Cancer Center

FibroGen limited access to Duchenne muscular dystrophy drug after failed clinical trial. But mom says it works for her son. - San Francisco Business Times

FDA panel: Benefit of 'highly anticipated' lung cancer drug can't be interpreted reliably
de
por adulto (o preço varia de acordo com o tamanho do grupo)







