ANANDA Scientific Announces FDA approval of the IND for the Clinical Trial on the Treatment of Opioid Use Disorder (OUD)
Por um escritor misterioso
Descrição
ANANDA Scientific Inc., (a biotech pharma company) today announced approval by the U.S. Food and Drug Administration (FDA) of the Investigational New

The Opioid Crisis and Recent Federal Policy Responses

Translating Kratom-Drug Interactions: From Bedside to Bench and Back

ANANDA Scientific and David Geffen School of Medicine UCLA Announce Clinical Trial Utilizing Liquid StructureTM Cannabidiol (CBD) for the Treatment of Opioid Use Disorder (OUD)

A Decade of FDA-Approved Drugs (2010–2019): Trends and Future Directions
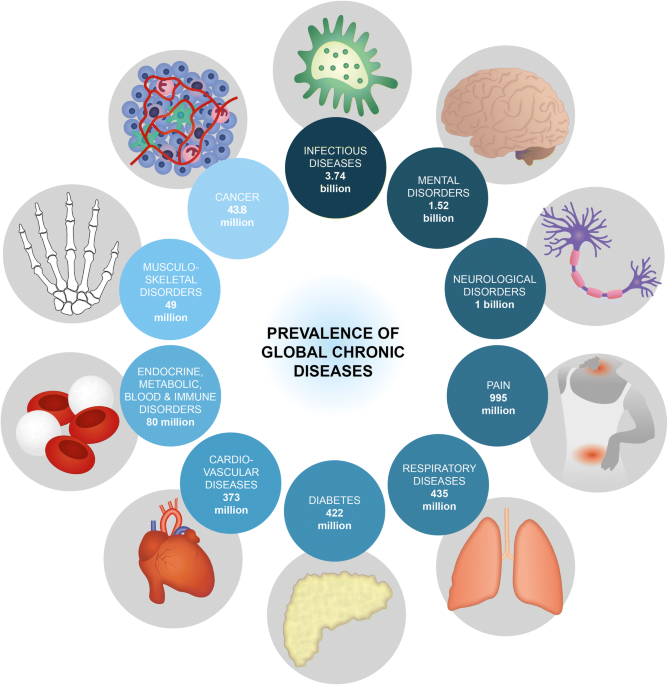
Advanced implantable drug delivery technologies: transforming the clinical landscape of therapeutics for chronic diseases

ANANDA Scientific

ANANDA Scientific
machine learning – NIH Director's Blog

g556911.jpg
de
por adulto (o preço varia de acordo com o tamanho do grupo)







